/chemical-periodic-table-of-elements-with-color-cells-vector-illustration-840464844-ff61af51081b4fb1959504105e232e01.jpg)
Using the above calculator you could find that e.g. Or 1 mole of a substance will contain Avogadro's number of that substance. The term " mole" is defined in that one mole of a substance with a molecular (or atomic) mass of one (1), will have a mass of 1 gram. It is defined to be 1/12 of the mass of one atom of carbon-12 and in older works is also abbreviated as "amu".Īlso, important in this field is Avogadro's number (N A) or Avogadro's constant (6.0221 x 10 23). In related terms, another unit of mass often used is Dalton (Da) or unified atomic mass unit (u) when describing atomic masses and molecular masses. Molecular mass or molar mass are used in stoichiometry calculations in chemistry. This Calculator has been tested on Internet Explorer version 6 only,įirefox might not show all fields correctly. For question or remarks please contact us. !!! Lenntech BV cannot be held responsible for errors in the calculation, Make sure you enter the molecule of crystallization at last (e.g. The calculator handles at most two different bracket levels. The molecular mass calculator will recognize the entered formula's, which are included in the list of organic compounds. Or you can choose by one of the next two option-lists, which contains a series of common organic compounds (including their chemical formula) and all the elements. This online calculator you can use for computing the average molecular weight (MW) of molecules by entering the chemical formulas (for example C3H4OH(COOH)3 ). Molecular Weight Calculator Molecular Weight Calculator
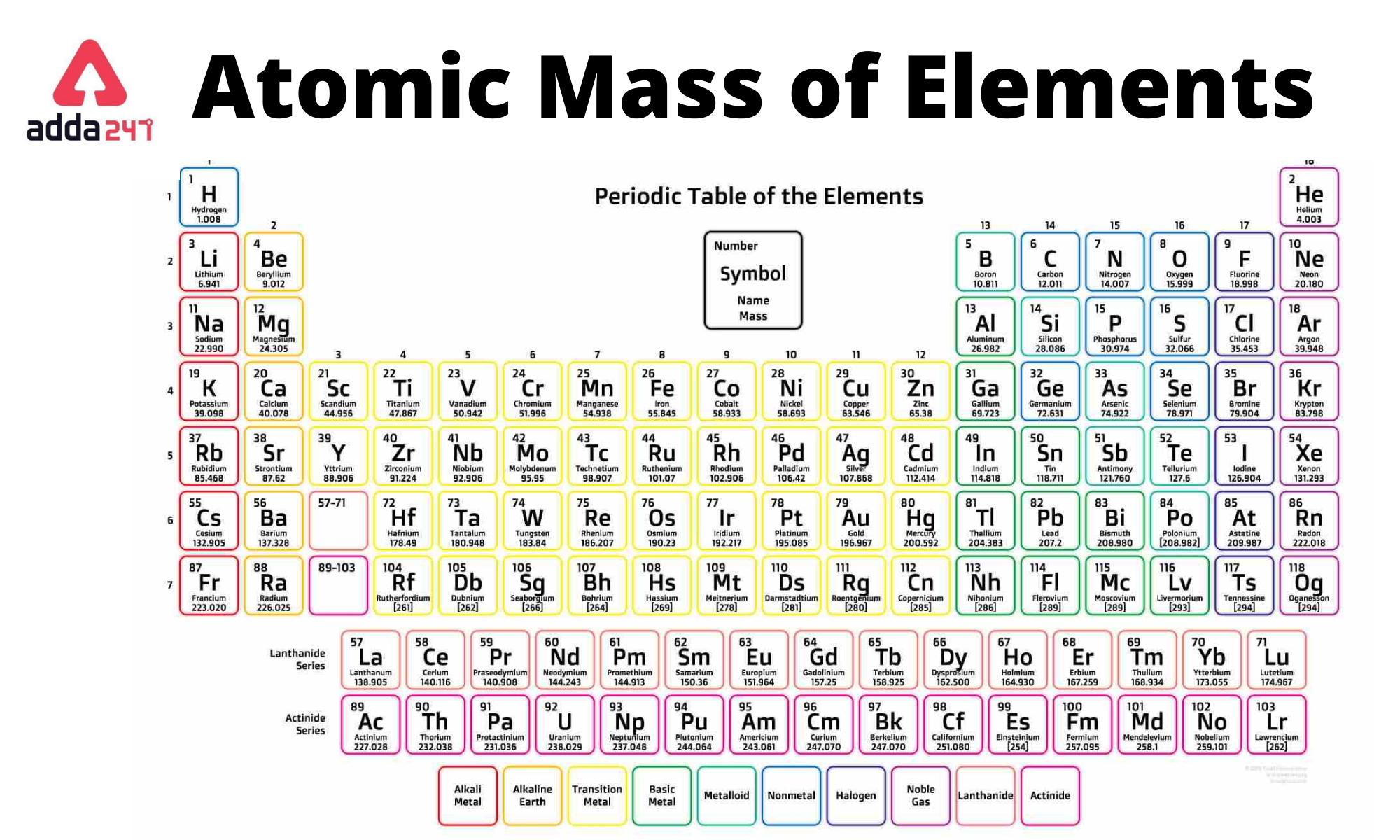
Separation and Concentration Purification Request.Plant Inspection & Process Optimalisation.Again, the mass of a mole substance is called the molar mass, and to find the molar mass of a substance you just need a periodic table and the chemical formula. That’s the total molar mass of the compound CO2. Then finally you add all products, so 12 plus 32 is 44. You would multiply it by its subscript of 2, which is 32. Then you’d do the same thing with oxygen. Then the way you would find that is first you would find the atomic mass of carbon, which is 12, and then you’d multiply it by the subscript, which is just an understood 1 right there, so the product there would be 12. If you were trying to find the molar mass of the compound carbon dioxide, which looks like this-CO2-and the molar mass is 44. Then, to find the molar mass of the compound, you’re going to add all those products together. By multiplying the atomic mass by the subscript you’re getting the product, you’re getting the molar mass product for that atom. That way, you’re finding the total molar mass for all of that atom because you’re finding the atomic mass, then you’re multiplying it by however many times it appears in the compound. After you’ve found the atomic mass, multiply the atomic mass by the subscript. You’re going to start with one atom at a time. I’m going to show you a 3-step process for finding the molar mass of a compound.įirst, find the atomic mass of an atom. You kind of follow the same protocol for finding the molar mass of an element, but then you combine all those molar masses together to find the molar mass of the compound. Now the molar mass of a compound can be calculated by summing the molar masses of each atom and the chemical formula. That’s how you find the molar mass of an element. Now we’ll write it with a different unit because it’s also our molar mass, and that will be grams over moles: \(\frac\).

If you’re looking at the element of copper you would look in the periodic table and find that it has an average atomic mass of 63.55 AMU, so that’s the molar mass. If you’re trying to find the molar mass of an element, then you’ll take the average atomic mass listed in the periodic table. To calculate the molar mass of a substance you need a periodic table and the chemical formula. The mass of a mole of substance is called the molar mass.
